The Trump administration is planning to “overwhelm” the airwaves with vaccine messaging by November, an HHS official told reporters last week.
But as the Trump administration gears up for an autumn messaging blitz, the mass, long-term studies of potential vaccines that the campaign will promote have only just begun. Meanwhile, President Trump’s political fortunes continue to lag.
Experts expressed concern to TPM that the Trump administration may be frontloading the public information campaign around deploying the vaccine with an eye toward the November election — and far in advance of when a vaccine would normally be ready.
The approach comes as vaccine researchers estimate that the two vaccine trials which began last week won’t inoculate a full cohort of participants until late September, and as those same researchers expect safety and efficiency data to be gathered through December at the earliest. A widely available vaccine would only come after that research is complete.
The messaging around the vaccine should “be very thoughtful about this and not overpromise,” Anna Durbin, a vaccine researcher at Johns Hopkins, told TPM, adding that manufacturing and distribution issues mean that widespread vaccination will likely be months away from federal approval of the first treatments.
“What you don’t want to have happen is have everyone in the country think they’re immediately going to get a vaccine,” she said. “Then, you’ve broken the trust that people have.”
As part of Operation Warp Speed, the Trump administration’s effort to turbocharge the development and deployment of a COVID-19 vaccine, the Department of Health and Human services has begun to review messaging strategies aimed at spreading awareness of the vaccine and motivating Americans to get inoculated against COVID-19.
One HHS official described the communications strategy on a call with reporters on Thursday as “much more truncated, time-wise,” and added that the department’s press secretary Michael Caputo would be helping with the effort.
“Think about it as a 4- to 6-week period of time, very intense, multi-channeled, highly targeted based on what we’ve learned about the vaccine,” said the official, who spoke with reporters on the condition of anonymity. “So, you may not hear a lot about promoting vaccines over the airwaves in August and September, but you’ll be overwhelmed by it come November.”
Emily Brunson, co-author of a July report from Johns Hopkins on messaging strategies around deploying a vaccine in the U.S., said that “the CDC should really be the spokesperson here and no one else.”
“The politicalization of health issues should never happen, and it’s going to cause additional hurdles for vaccination,” she added.
Creating a vaccine
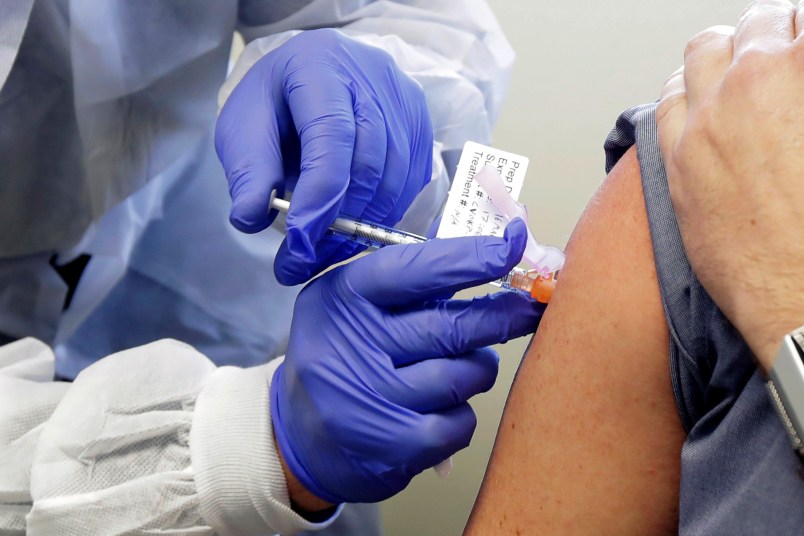
Two vaccine trials — one developed by Pfizer and the other by Moderna — have started this month in the U.S.
The tests are being run on 30,000 people each, and constitute live experiments designed to help develop a safe and effective vaccine during a pandemic.
“There’s a lot of thinking that we’re in a pandemic, and so we need to throw out the usual rule-book,” said Zoe McLaren, an associate professor of public policy at the University of Maryland, Baltimore County. “But the trial itself is designed to happen during a pandemic, and to be responsive to the needs of a country during a pandemic.”
It takes between six and eight weeks to gather subjects and inject them with the vaccine, making late September the earliest point at which the trials would have full enrollment.
Subjects are chosen in part by their proximity to COVID-19 hotspots, allowing researchers to see more quickly whether the portion of subjects who receive a real vaccine, and not a placebo, achieve immunity or experience weaker infections.
“The individuals that receive the vaccine have to be exposed naturally to it,” Dr. Susan Hassig, an epidemiologist at Tulane University, told TPM. “Even in our most heavily affected environments, maybe one percent of people might be infected at any one point in time — it’s not going to be instantaneous exposure.”
The vaccine’s effectiveness can only be determined once enough people in the placebo group have been infected with COVID-19 to see if there’s a meaningfully lower infection rate in the vaccinated group.
That threshold — usually a set number of cases in the group needed to show whether the vaccine works — can be adjusted downwards in emergencies like a pandemic to facilitate quicker approval, though at increased risk.
Durbin, the Johns Hopkins vaccinologist, described the threshold as a “trigger for an analysis” before the FDA issues an emergency use authorization that could enable initial access to the vaccine, likely given first to frontline health-care workers.
She added that, by November at the earliest, researchers would like have a better idea “of what the landscape looks like, and possibly if they’ll be able to ID a vaccine by the end of the year.”
Uncertain timeline
Experts stressed that any timeline related to a vaccine was extremely uncertain, and that public health campaigns around deploying vaccines often come down to the details of how those vaccines work.
The risks of touting a vaccine too early, however, are immense. Messaging campaigns around vaccination are based on the details of the vaccine and the populations they’re meant to treat.
The HHS official emphasized that the campaign would be “highly targeted” based on data from the vaccine trials, though it’s unclear how much could be known about the various vaccines’ effectiveness by that time.
A single-dose vaccine that is effective on many groups of people, but not the elderly, for example, would require a different messaging campaign than a vaccine which required two doses a month apart, but which remained effective across different groups of people.
“One of the problems around doing it too early is you don’t know what the vaccine is,” said Durbin, the Johns Hopkins researcher. “We’re all hopeful, but you don’t want to promise something that never shows up.”






There are only two things we need to remember.
Eye roll maybe?
People are already dying so if an untested vaccine kills people what is the problem?
An untested, reelection-driven, vaccine pushed by a raving lunatic?
What could possibly go wrong with that?
Of course he will. The only question is which pharma company/companies will be willing to take Trump’s multi-billion dollar payoff.
Dr. Birx? This will be your last chance to save yourself.