WASHINGTON (AP) — The nation’s first COVID-19 vaccine will begin arriving in states Monday morning, U.S. officials said Saturday, after the government gave the final go-ahead to the shots needed to end an outbreak that has killed nearly 300,000 Americans.
Trucks will roll out Sunday morning as shipping companies UPS and FedEx begin delivering Pfizer’s vaccine to nearly 150 distribution centers across the states, said Army Gen. Gustave Perna of Operation Warp Speed, the Trump administration’s vaccine development program. An additional 450 or so facilities will get the vaccine between Tuesday and Wednesday.
Initially, about 3 million shots are expected to be shipped nationwide. It was unclear exactly who would receive the first doses of the vaccine, though health workers and nursing home residents were the priority. Perna said health authorities would decide.
A similar number of shots will be held back for those recipients’ second dose, which is needed for full protection from COVID-19.
The announcement Saturday kicks off a massive logistical operation involving the federal and state governments, private companies and health care workers to quickly distribute limited vaccine supplies throughout the U.S.
Perna compared the effort to D-Day, the U.S.-led military offensive that turned the tide in World War II.
“D-Day was the beginning of the end and that’s where we are today,” Perna said a news conference. But he added that it would take months of work and “diligence, courage and strength to eventually achieve victory.”
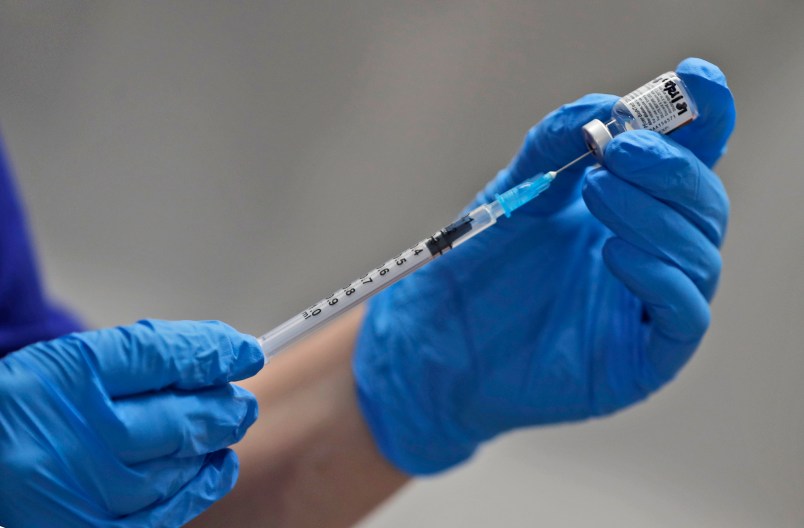
The first shipments are expected to leave Pfizer’s manufacturing plant in Kalamazoo, Michigan, via truck and then be flown to regional hubs around the country. Medical distributor McKesson and pharmacy chains including CVS and Rite-Aid also are involved in local rollout.
In a key distribution challenge, the vaccine, co-developed with BioNTech, must be stored and shipped at ultra-low temperatures of about 94 degrees below zero. Pfizer has developed shipping containers that use dry ice, and GPS-enabled sensors will allow the company to track each shipment and ensure it stays cold.
Distribution locations include hospitals and other sites able to meet those ultra-cold storage requirements. Within three weeks, vaccines should be delivered to all vaccination sites identified by state governments, such as local pharmacies, Perna said.
The vaccine was timed to arrive Monday morning so that health workers would be available to receive the shots and begin giving them, Perna said.
It comes after the Food and Drug Administration late Friday authorized emergency use of the vaccine. The signoff capped an unprecedented global race to speed vaccines through testing and review, chopping years off the normal development process.
The FDA found the vaccine highly protective with no major safety issues. U.S. regulators worked for months to emphasize the rigor and independence of their review, but the Trump administration pressured the agency up until the final announcement. A top White House official even threatened to remove FDA chief Stephen Hahn if a ruling did not come before Saturday.
Concerns that a shot was rushed out could undermine vaccination efforts in a country with deeply ingrained skepticism about vaccines. Hahn again emphasized his agency’s independence to reporters Saturday.
“Science and data guided the FDA’s decision,” Hahn said. “We worked quickly because of the urgency of this pandemic, not because of any other external pressure.”
While determined to be safe, regulators in the U.K. are investigating several severe allergic reactions. The FDA’s instructions tell providers not give it to those with a known history of severe allergic reactions to any of its ingredients.
The FDA’s vaccine director, Dr. Peter Marks, said the agency will carefully track any reports of allergic reactions in the U.S.
“I think we still need to learn more, and that’s why we’ll be taking precautions,” Marks said.
The FDA next week will review a second vaccine from Moderna and the National Institutes of Health that appears about as protective as Pfizer’s shot. On Friday, the Trump administration announced it had purchased 100 million more doses of that vaccine on top of 100 million it previously ordered.
The announcement came after revelations last week that the White House opted not to lock in an additional 100 million doses of Pfizer’s vaccine for delivery in the second quarter of 2021. The Trump administration contends the current orders plus those in the pipeline will be enough to accommodate any American who wants to be vaccinated by the end of the second quarter of 2021.
___
Associated Press Medical Writer Lauran Neergaard in Alexandria, Virginia and writer Candice Choi in New York contributed to this story.






Here’s hoping the WH doesn’t try to interfere with where the deliveries take place. Between now an next week, anything is possible.
This is a promising development, but it’s a tiny bit of what the nation needs, and it’d clear the Trump administration has not planned out how to distribute this to everyone. Getting it to medical personnel is critically important though, we need every single one with the path that CIVOD is tearing through the people, every doctor or nurse that falls ill or dies means many more sick and dead Americans who they would have helped.
I’m hoping that the people doing this job manage to get past the Trump administration incompetence, and that the incoming Biden administration is able to handle the virus response and vaccinations the way it should have been done from the beginning. We still have at least six months before things will really be better, and we can start to live more safely, but it’s likely now going to take until 2022 before this is all over, and even then we won’t go back to normal but find a new normal instead.
I’m just waiting for the anti-vaxxer deplorebels to storm the vaccination sites.
Gotta fight the hoax that stole the election, right?
OT the Wisconsin Federal suit was dismissed with prejudice by the Trump appointed judge.
back in the day Birx still had some respect her correspondence with the governor here was explained as communication…
the estimated delivery here now for Category 1A won’t cover half of the health care workers, with none of the nursing homes supplied… and rumor is the original number is being cut
reagents used in vaccine production are gone, used up in the recipe for the testing program…
hiccups? I hope it’s not strangulation.