Historically, anti-abortion litigants have often been the ones to try out novel legal arguments to further the movement and make the procedure more difficult to obtain.
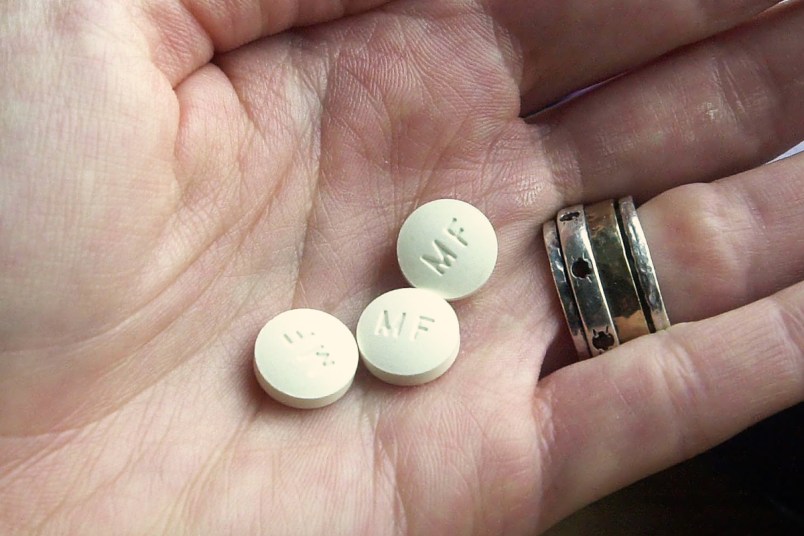
In West Virginia, the roles were reversed. GenBioPro, the maker of generic mifepristone, sued in January under the Supremacy and Commerce Clauses, arguing that the state’s near-total abortion ban was preempted by the Food and Drug Administration’s increasingly lenient restrictions around mifepristone.
On Thursday, Judge Robert Chambers, a Bill Clinton appointee, rejected most of GenBioPro’s arguments — though he did find that the state’s ban on prescribing mifepristone via telemedicine is in direct contradiction with the FDA’s parameters, and sustained the lawsuit on that count.
He granted the motion for dismissal from West Virginia officials on the constitutional clause arguments.
“Here, West Virginia’s UCPA has limited when an abortion may be performed, without touching how medication abortion is to be performed,” he wrote, referring to the abortion ban, the Unborn Child Protection Act. “The mifepristone [restrictions] only concern themselves with the latter.”
Chambers did take a swing at the West Virginia officials’ attempt to decree the FDA’s actions subject to the major questions doctrine, an increasingly in-vogue right-wing legal theory that demands that every agency action of political or economic significance be authorized with a very specific congressional statute. (It has, in recent years, been used by the conservative Supreme Court majority to knock down regulations on everything from climate change-causing greenhouse gas emissions to student loans.)
Chambers got to the heart of the lengths to which anti-agency actors try to stretch this theory to cover all regulation they don’t like.
“Defendant misunderstands the purpose and scope of the statutory grants of agency authority by demanding that Congress have listed every possible medical condition and procedure when it instructed the FDA to regulate prescription medicine generally,” he wrote. “For example — imagine if Congress were forced to list every endangered species for the Endangered Species Act (“ESA”) to grant the Fish and Wildlife Service (“FWS”) authority to protect any specific at-risk organism.”
“Calling this a ‘major questions case’ and demanding the FDA refrain from treating abortion medications on par with other medications under the FDCA would make just as much sense as demanding the FWS refrain from listing the snail darter as an endangered species under the ESA,” he added with a flourish.
The West Virginia case is one of many centered on abortion medication currently wending their way through the federal court system.
The Fifth Circuit just issued a decision that would reimpose a raft of restrictions the FDA has lifted from mifepristone, though that decision remains on ice until the Supreme Court rules.
Another case is slowly making its way through federal court in Washington, where Democratic attorneys general are fighting to lift the current restrictions on mifepristone.
The case at the Fifth Circuit, and maybe more of these, will almost certainly reach the Supreme Court. In the post-Dobbs landscape, much of the litigation frenzy has focused on mifepristone, the availability of which through the mail has provided relief even for women living under the most draconian anti-abortion regimes.
Read the West Virginia ruling here:






Dumb idea.
Government doesn’t work to the degree Republicans impose their dumb ideas.
Frist!!
The RWNJ’s trying to keep Women from getting mifepristone. GLWTS. The entire History of Prohibition and the legalization of Marijuana demonstrates a principle grounded in fact if not in law:
For GOP judges, it is often not the logic, or logical consistency, that is important in rulings, any more than longstanding precedent. What matters is consistency with the GOP’s current political requirements, and making rulings sound as if they are consistent with “tradition”, regardless of how radical the outcomes really are.
I think Judge Chambers missed the fact that the anti-regulatory nuts do in fact want Congress to have to declare that the snail darter is an endangered species and requires protection. The idea that Congress would say “an endangered species as defined by science should be protected” and then the executive would lay out how that works (based in the science) is not what they want…mostly because they want to block the government from doing anything helpful as they know they can gridlock Congress to stop the many required detailed bills but not the more general ones (which are already on the books).
Well, except when it’s their agenda that they want, then they are fine with the executive pushing the agenda, without regard to the rules or laws. Dictatorship instead of democracy is their eventual goal, they just want to make sure the government can’t stop them or help people before they manage the takeover.
Team Unicorn…the curse that keeps on giving…